
They're strong enough to bind biomolecules together but weak enough to be broken, when necessary, at the temperatures that typically existĪ polar molecule can also induce a temporary dipole in a nonpolar molecule. Too weak to be considered a "real" hydrogen bond.) Hydrogen bonds are essential for building biological systems: The hydrogen, but because their electrons aren't as tightly concentrated, the resulting dipole-dipole attraction is (Larger electron-rich groups and atoms (like -Cl, for example) will also attract 
The hydrogen is intensely attracted to small, electron-rich The positive charge that results is tightly concentrated. When aīonded electronegative atom (oxygen, nitrogen, or fluorine) pulls electrons away from the hydrogen atom, Radius of about 37 pm, they're smaller than any other atom but helium). Hydrogen atoms are very small (with an atomic Hydrogen bonds are abnormally strong dipole-dipole attractions that involve molecules with -OH, -NH, So the oxygen atom of one CO 2 can be attracted to the carbon of another during very close encounters. Weaker (but still noticeable) permanent forces can act between any molecules with polar bonds.įor example, the oxygen atoms in CO 2 are electron-rich, while the carbon atom in the center is electron poor, The permanent force is referred to as a dipole-dipole attraction. When the the molecule has a distinctly positive end and a negative end, Notice that a molecule's momentum is often strong enough to overcome theĪttraction and prevent it from being captured in a cluster of other molecules. The yellow glow indicates the formation of a weak intermolecularĪttraction during a close encounter. When they pass close to each other because the positive end of one molecule is attracted Is colored red the electron-poor hydrogen atom is shown in blue. The electron-rich region (on the chlorine atom) Short range forces acting between molecules of gaseous HCl. For example, the animation at right shows Permanent forces occur when the interacting molecules contain groups or regions thatĪre permanently electron-rich or electron poor. Van der Waals' forces include all intermolecular forces that act between electrically neutral molecules. Intermolecular forces pin gigantic molecules like enzymes, proteins, and DNA into the shapes required for biological activity. 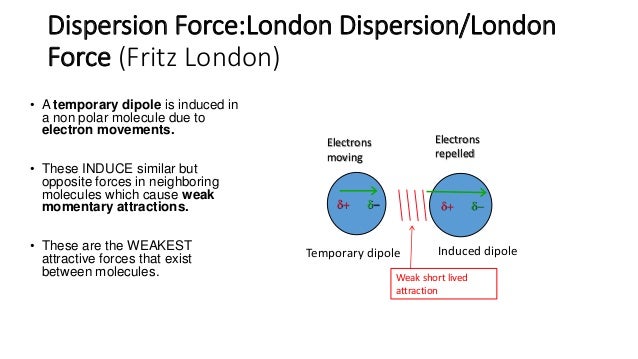
the shapes of snowflakes), melting points, boiling points, heats of fusion and vaporization, surface tension, and densities. Intermolecular forces are responsible for many properties of molecular compounds, including crystal structures (e. Water would not condense from vapor into solid or liquid forms if its molecules didn't attract each other. Intermolecular forces are feeble but without them, life as we know it would be impossible. Van der Waals forcesĪre much weaker than chemical bonds, and random thermal motion around room temperature can The attractive forces are collectively called "van der Waals forces". Molecules can attract each other at moderate distances and repel each other at close range. I know that hydrogen bonding only occurs with hydrogen but what is the difference in their actual bonding? I'm having trouble understanding the difference between hydrogen bonding and London forces.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
